Fermented / Cured Meats
Validation
Fermentation, drying, and smoking meats are probably the oldest forms of meat preservation. These two processes are mentioned together because, in practice, they are almost inseparable. These preservation methods are at least several thousands of years old, even being referenced in Homer's Odyssey!
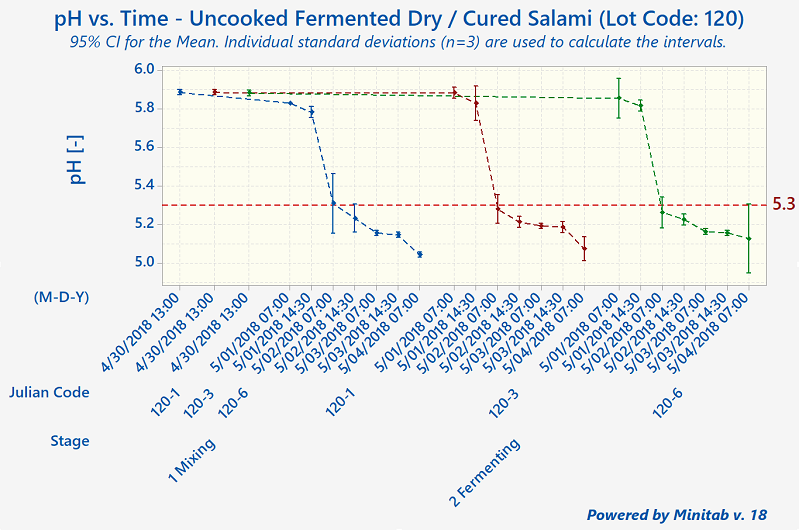
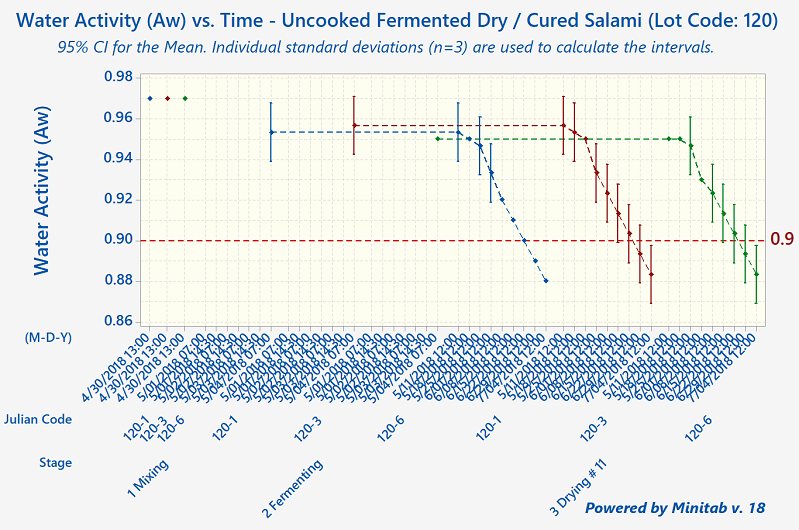
Since that time, humans have come to understand that, although these processes preserve meats well, they aren't foolproof. The modern-day validation process for uncooked fermented and dry-cured meats involves the collection of a lot of process data, including pH, Aw-value, temperature, relative humidity, and degree-hours).
Quantum Food Solutions works with companies to establish successful validation studies that meet their needs. Our food safety consultants can help your company come up with detailed descriptions of the worst-case-scenarios for the product or products under evaluation. These are based on factors such as grind size, calibre, starter culture, substrate, initial microbial load from the ingredients, cold-spots in fermentation rooms (the zone where a product has the slowest fermentation rate or pH drop), and "wettest-spots" in drying rooms (slowest drying rates).
Exporting These Products To The United States
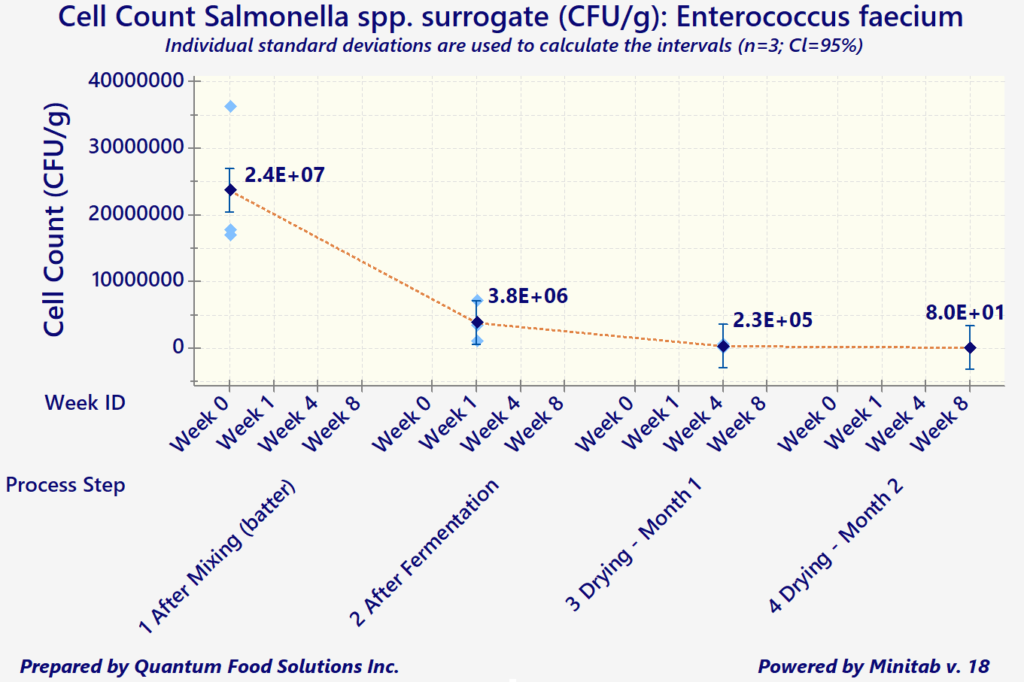
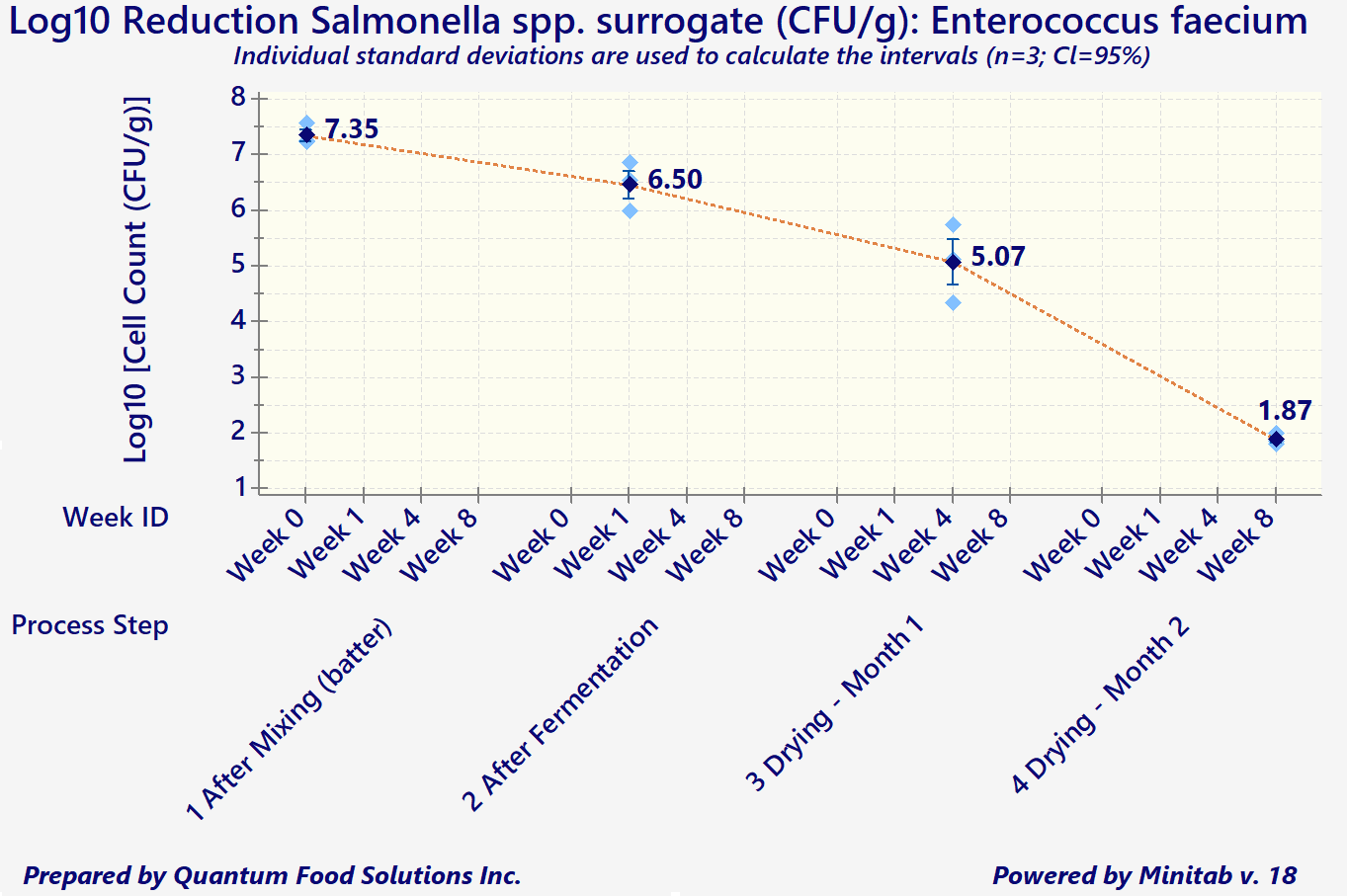
In addition to the above considerations, any processors wishing to export to the US are required to demonstrate that the existing process is capable of attaining a minimum 5-Log reduction in both Salmonella spp., and E. coli O157: H7 (for products containing beef).
"In-Plant" data should be collected through an inoculation study, per FSIS HACCP Validation Guidelines. However, since a pathogen such as Salmonella would be too dangerous to be introduced into any meat processing plant, the use of surrogate organisms is highly recommended and is also found acceptable by FSIS.

Hazards, Control Measures, And 5-Log Reduction?
Log reduction measures the thoroughness of a decontamination process and how it reduces the concentration of a contaminant. It's defined as the common logarithm of the ratio of the levels of contamination before and after decontamination. An increment of 1 corresponds to a reduction of the contaminant concentration by a factor of 10. The ‘5-Logs’ is the difference of the exponents (base 10) between the initial and final cell counts/gram of product.
Under the current regulatory framework, the following hazards have been identified as a public concern:
Salmonella spp., & E. coli O157: H7 (products containing beef)
Under Canadian regulations, processors are allowed to use pH and Aw-value (independently or in combination). There is no actual ‘prescribed’ regulatory standard such as a 5-Log reduction. However, the 5-Log reduction does become a regulatory performance standard required for exports to the United States.
Listeria monocytogenes
Under Canadian regulations (Health Canada’s Policy on Listeria monocytogenes in RTE Foods), processors are allowed to sell these products provided that the product does not exceed a count greater than 100 CFUs/g during its usable shelf-life. This is monitored regularly through CFIA’s mandated testing of RTE products (i.e., CFIA Category 2B).
As a simple example, let's say an initial product batter starts with approximately 1,000,000 cells per gram (10^6 CFUs/g). The combination of product composition and acceptable processing parameters must decrease that number to 10 cells/gram (or 10^1 CFUs/g) by the end of the process (for meat, often it's drying).
If you need help coming up with a plan for your meat processing facility, Quantum Food Solutions uses scientific and engineering expertise to come up with a custom food safety plan.